Introduction
Homeopathy is a complementary health practice that often stimulates lively discussion among its proponents and skeptics. The debate stems from the use of ultra-diluted homeopathic medicines, which many skeptics believe contain no active ingredients, leading critics to attribute any therapeutic benefits to the placebo effect. Proponents of homeopathy, however, believe that it is a holistic system of medicine that works to support the body’s innate healing abilities.
Homeopathy was established in 1796 by a German physician named Samuel Hahnemann, who later published the first complete textbook in 1810.1-2 In over two hundred years since it was introduced, worldwide use of homeopathic medicines continues to expand, with the market projected to grow from $15.96 billion in 2025 to $28.81 billion in 2034 reflecting a compound annual growth rate of 6.78% from 2025 to 2034.3-8
The growing global use of homeopathic products presents an opportunity for pharmacy professionals to expand their knowledge of homeopathy to meet increasing consumer demand for information and ensure its safe use. This article aims to introduce scientific foundations of homeopathy and provide pharmacy professionals with the key educational insights needed to effectively counsel patients and address consumer questions about this complementary therapy.
History of Homeopathy
Homeopathy was developed based on the idea of “similia similibus curentur”, Latin for “let like be cured by like.”1-2 After recognizing the inadequacies of standard medical practice during the 18th century, Samuel Hahnemann stepped away from the medical profession to translate scientific literature. While translating the work of a Scottish physician, William Cullen, Hahnemann studied Cinchona officinalis, the Peruvian Bark from which quinine is derived and a widely used treatment for malaria at the time. Hahnemann was not satisfied with Cullen’s description of Cinchona’s action in treating malaria, so he embarked on a journey of experimentation that ultimately led him to formulate the principle of similarity.1-2
Upon ingesting a high dose of Cinchona while healthy, he experienced intermittent fever, chills, and fatigue, symptoms that are similar to those of malaria. From this, he explained that a substance that could elicit symptoms in a healthy person is capable of curing the disease in the sick patient experiencing similar symptoms. This observation led Hahnemann to formulate the Law of Similars, based on the principle that “like cures like.”1-2
As Hahnemann applied the Law of Similars into practice, he found that giving standard doses to patients often led to a temporary worsening of symptoms before any clinical improvement was observed. To lessen these initial aggravations, he found that lowering the doses reduced adverse reactions while still preserving the therapeutic benefits. Hahnemann proposed that a substance that can produce symptoms in a healthy person may relieve similar symptoms in an ill patient when administered in extreme small doses. Hahnemann continued to experiment with these ideas while developing the potentization process, combining the serial dilutions with vigorous shaking, which was believed to release the healing properties of the substance.1-2
Hahnemann had established homeopathy based on two central principles: the Law of Similars and the Law of Minimum Dose, asserting that a substance which is capable of causing specific symptoms in a healthy person can, in extreme low dose, treat those same or similar symptoms in an ill individual.1-2
Homeopathy and Other Complementary Therapies
Hahnemann was credited with coining the terms allopathy, based on the idea of “contraria contrariis” or “opposites oppose”, and homeopathy based on the notion of “let like be cured by like.”1 Today, allopathy refers to the use of conventional medical treatments to manage disease, whereas homeopathy is a system of medicine used as complementary therapy.1
The terms “complementary”, “alternative”, and “integrative” therapies are often used to refer to non-allopathic approaches to health and wellness. The National Center for Complementary and Integrative Health (NCCIH) defines complementary health as wellness practices used in combination with conventional (allopathic) medicines, whereas using these approaches instead of conventional treatments is considered alternative. Integrative health incorporates both conventional and complementary therapies in such a way to treat the whole person rather than one organ system.9
Complementary health practices are categorized by how they are taken by the individual. Mind and body practices include those that are physical (e.g. massage), psychological (e.g. mindfulness, spiritual practice), or combination of both (e.g. acupuncture, meditation, tai chi, yoga, among others). Natural products encompass nutritional products which are mostly marketed as dietary supplements (e.g. vitamins, minerals, herbal remedies, probiotics, amino acids, and others).9
Homeopathic medicines differ from dietary supplements in their therapeutic principles. While homeopathy is based on the principles of similars and minimum dose, treatment with dietary supplements is based on the conventional pharmacological principle in which the active ingredient must reach a therapeutic concentration to exert its mechanism of action and produce a response.1,10
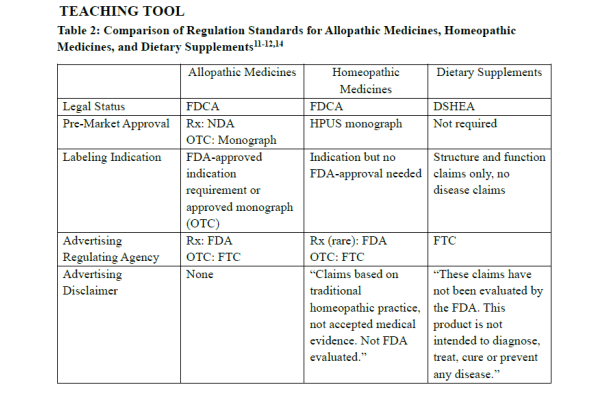
Although homeopathic medicines are classified as complementary therapy, they are often incorrectly grouped with dietary supplements and mistaken as herbal remedies.9 Dietary supplements contain ingredients intended to complement the diet (e.g. vitamin, mineral, herbs, amino acids, probiotics, and others) and are regulated as food.9,11 Homeopathic medicines, which are derived from plants, minerals, or animals, are regulated as drugs. As such, homeopathic products and dietary supplements have different federal regulatory standards.11-14
One key regulatory distinction between homeopathic medicines and dietary supplements lies in their product labeling. Homeopathic product labels must include their purpose or use for specific symptoms or conditions, active ingredients name with potency dilutions, and the “Homeopathic” designation. In addition, homeopathic medicines may make limited therapeutic claims to symptom relief; whereas dietary supplements are prohibited from making claims to diagnose, treat, cure, or prevent a disease. Dietary supplement product labels are restricted to structure or function claims only.11-14
Homeopathic Medicine Manufacturing
Source Materials
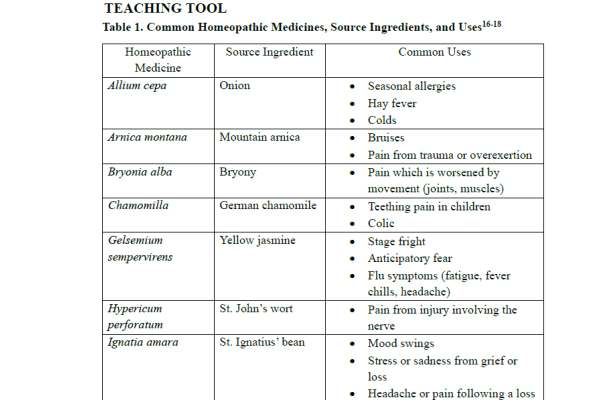
Homeopathic medicines are produced from plants, minerals, or animal-derived ingredients. Most common homeopathic medicines come from plant sources.15-16 Examples of plant-derived homeopathic medicines include Arnica montana (Mountain Arnica, also known as the Leopard’s Bane), Allium cepa (red onion), Ignatia amara (St. Ignatius’ bean), Gelsemium sempervirens (yellow jasmine), Nux vomica (poison nut), Rhus toxicodendron (poison ivy), and Hypericum perforatum (St. John’s wort).12-13 Table 1 lists some common plant-sourced homeopathic medicines and their uses.16-18
Mineral and animal-derived extracts are also found in several other homeopathic medicines. Examples of mineral-sourced products include Calcarea carbonica (calcium carbonate) and Natrum muriatium (sodium chloride), while animal-derived homeopathic medicines include Apis mellifica, (honeybee), Cantharis vesicatoria (Spanish fly), and Lachesis mutus (venom of the bushmaster snake).15-16
There are over 300 homeopathic medicines cataloged in the Materia Medica, and about one-quarter of them are listed for practical use on the National Center for Homeopathy “Homeopathy Remedies Materia Medica” tool.15-16
Potentization Process
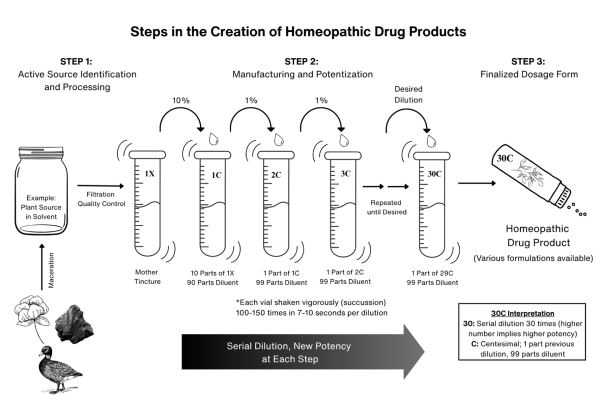
The creation of homeopathic products involves the identification and preparation of the natural source, the potentization process, and the production of the desired preparation form. The first step in homeopathic medicine creation involves the identification and processing of source materials. Adhering to established good manufacturing practice guidelines and quality control standards, the raw plant or animal-based ingredients are macerated in diluents of pure water and alcohol mixture, while the minerals are triturated. Thereafter, the soluble materials continue to soak, allowing the active ingredients to dissolve to produce the original extract from which dilutions are prepared, including the mother tincture (MT). The MT is then separated and stored in an environment controlled for light, temperature, and ventilation until it is ready for the potentization process.19
Potentization is a unique process in homeopathy in which the MT undergoes serial dilution followed by vigorous shaking, a step known as succussion.1,14 Homeopathic potencies are produced from the MT through serial dilutions using the standardized scales, most commonly the Centesimal (C) and Decimal (X) scales. Both dilution scales use similar potentization processes involving serial dilution and succussion; however, they differ in their solute-to-solvent ratios.1,19
Centesimal dilution uses the 1:100 ratio scale, where 1 part of the 1C potency is diluted in 99 parts of solvent to create the 2C potency. After which, the dilution is succussed. In comparison, Decimal dilution utilizes the 1:10 ratio scale, where 1 part of the 1X potency is diluted in 9 parts of solvent to form the 2X potency, followed by succussion.1,19-20
For example, most plant-derived MTs are 1X because they are made with 1 part of the plant source and 9 parts of solvent (usually a mixture of pure water and alcohol). To prepare the 1C potency, the 1X MT is diluted by a factor of 10 (1 part MT: 9 parts solvent). Thereafter, the 1C potency is mixed with 99 parts of the diluent and succussed forming the 2C potency. This process is repeated using the same 1:100 ratio (1 part of the 2C and 99 parts of diluent), followed by succussion to achieve the subsequent 3C potency.1,19-20 Figure 1 illustrates the steps involved in the making of a plant-derived homeopathic drug product.
The final step in the manufacturing process involves formulating the dilutions into various preparations including sublingual pellets, tablets, oral liquids, topical ointments, creams, gels, sprays, and suppositories.1,19
Homeopathic products for oral administration are commonly available on U.S. market in Centesimal (6C and 30C) or Decimal (6X or 30X) potencies; and topical products are mostly available in Decimal potencies, usually ranging from 1X to 3X potencies, which contain low dilutions of the active constituents from the source material.16-18 Homeopathic medicines are available either as individualized single medicines or as multi-ingredient combinations marketed as branded products.1,21-23
Homeopathy Regulatory Framework
Classified as drugs by the Federal Food, Drugs, and Cosmetic Act (FDCA) of 1938, homeopathic medicines are regulated as such and are primarily marketed in the U.S. as over the counter (OTC) products.12-13
The classification of homeopathic products as drugs places its regulatory authority under the U.S. Food and Drug Administration (FDA). Therefore, homeopathic drug products (HDPs) must comply with product quality and Good Manufacturing Practices (GMP), including their manufacturing, processing, packing, labeling, storage, and handling.12 The FDA recognizes the Homeopathic Pharmacopoeia of the United States (HPUS) as the official compendium or the legal reference source for about 1300 homeopathic active ingredients listed in its monographs.24 Additionally, the HPUS also contains manufacturing protocols for HDPs.24
Although classified as drugs, HDPs differ from conventional allopathic medications in that they are not subject to FDA premarket approval, which means HDPs are not required to undergo the new drug application (NDA) process involving clinical trials to show safety and efficacy prior to marketing.12-13 Instead, they are accepted for inclusion as monographs by the HPUS, which is used to establish standards for identity and preparation of HDPs.12,24 While not an FDA approval process, HPUS monograph submission requires toxicology and therapeutic use data for review by the committee which includes experts in medicines, biology, chemistry, botany, pharmacy, and others, to ensure standardization and safety.24
In 2017, the FDA introduced an industry draft guidance which proposed six risk-based enforcement priorities for HDPs, with increased oversight into targeted areas that present the greatest potential risk, including:12
- Products with reported safety concerns
- Products that contain ingredients associated with potential safety concerns
- Products other than those taken orally or topically
- Products for vulnerable populations (e.g. infants and children, the elderly, pregnant women, and immunocompromised people)
- Products that do not meet standards for quality
- Products intended for prevention or treatment of serious and/or life-threatening diseases or conditions.
By December 2022, the Homeopathic Drug Products Guidance for FDA Staff and Industry was finalized and targeted enforcement of risk-based regulation was implemented.12
In addition to FDA regulations, the Federal Trade Commission (FTC) is another governmental agency that regulates advertising and marketing practices of HDPs, which require advertising claims to be truthful, not misleading, and substantiated.25-26 Homeopathic product labels must include the same essential components as allopathic drug labels including active ingredients with their purpose, uses, warnings, directions, storage information, and inactive ingredients. The active ingredient name is followed by the potency (e.g. 6C, 30C) and the notation “HPUS”, which is an indicator denoting that the ingredients are monographed in the official compendium. In addition, homeopathic product labels must carry the disclaimer: “Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.”25-27
Table 2 provides a summary of regulation requirements for allopathic medications (prescription and OTC), homeopathic medicines, and dietary supplements.11-12,14
Homeopathy Global Use
Worldwide survey data indicates the prevalence of homeopathy use is estimated to range between 0.7% – 9.8%, averaging about 3.9% in many European and Asian countries, Australia, Canada, Israel, and the U.S. Of the reported countries, those that had the highest prevalence of use were countries where homeopathy is a required part of health insurance coverage.3 In India, homeopathy is widely accepted and recognized as a medical system that is represented in public health under a central government ministry. With over 280,000 registered practitioners, homeopathy centers encounter about one-fifth of the patient load as allopathic medical centers despite being outnumbered 19 to 1.4
The 2012 U.S. National Health Interview Survey (NHIS) reported an annual prevalence rate of homeopathy use to be about 2.2% among American adults, accounting for an estimated five million adults and one million children.5-7 The same survey reported homeopathy use significantly increased by 15% compared to prior surveys. The most common conditions for homeopathy use were related to symptoms associated with respiratory, ears, nose, and throat, and musculoskeletal pain.5-7
Selection of Homeopathic Medicines
Treatment choice is guided by the distinction between individualized homeopathic medicine (IHM) and non-individualized homeopathic medicine (non-IHM).1 In practicing IHM, the focus is on the person rather than the disease. Therefore, product selection requires examining the patient’s “symptom totality” and tailoring the treatment to the individual with usually a single medicine.1 The approach to IHM is to emphasize not only on the primary complaint but also the patient’s unique symptom etiology, specific experienced sensations, the modalities or factors that worsen or improve the symptoms, and concomitant symptoms or those occurring concurrently to the primary symptom complaint (e.g. “what else is this person experiencing?”).15 Meanwhile the practice of non-IHM focuses on the specific disease pathology rather than the patient’s individualized characteristics, with the treatment approach that includes multiple homeopathic medicines to manage the clinical symptoms.1
Homeopathy is a complementary health approach that focuses not only on the principles of similars and microdosing but also on individualization to the person. Because homeopathic medicines are available mostly as OTC products, individuals may utilize self-care treatments for minor illnesses. However, homeopathic practitioners may be consulted for an individualized approach that aims to promote overall health and wellness to the specific person.28 The American Institute of Homeopathy is one of several organizations which has a directory of homeopathic practitioners to provide consultations and individualized care.29
Research in Homeopathy
The current state of homeopathy research includes physicochemical, biological, and clinical studies. Physicochemical and biological research aims to address homeopathy’s mechanism of action through proposed constructs at the molecular and cellular level, while clinical trials focus on demonstrating safety and effectiveness in humans.
Physicochemical models provide a framework for experimentation in the transfer and retention of medicinal properties in highly diluted homeopathic medicines. Meanwhile, one of the biological models is based on the hormesis construct which states that a substance that is toxic at high doses may, when given in an extreme low dose, stimulate an organism’s natural adaptive self-regulatory responses.30 Future research aims to combine scientific concepts involving the hormesis biological model to that of nanoparticle physicochemical concepts to explain how highly diluted homeopathic medicines might interact and elicit the body’s natural healing responses.31
From the clinical research perspective, many randomized placebo-controlled trials have reported positive treatment effects with homeopathy when compared to placebo.32 In a randomized study published in 2024, investigators conducted a double-blind, placebo-controlled feasibility trial to evaluate the effects of IHMs in patients with knee osteoarthritis. Forty participants were assigned to receive either IHMs or identical placebos for two months, in addition to standardized care. Compared to placebo, the IHM group showed statistically significant improvements across the Knee Injury and Osteoarthritis Outcome Score (KOOS) subscales, including pain (p=0.010), stiffness symptoms (p=0.015), activities of daily living (p=0.032), sports and recreation function (p=0.006), and quality of life (p=0.026). Most frequently used homeopathic medicines were Sulphur, Arnica, and Natrum muriaticum. No serious adverse events occurred. These findings suggest that IHMs may improve outcomes in knee osteoarthritis, though larger trials are needed to confirm efficacy.33
Despite published trials reporting positive results, limitations to randomized clinical trials exist.34-35 Study design constraints including small sample size, variable treatment interventions, lack of control for confounding variables, inconsistent outcome endpoints, and lack of safety reporting have led to critiques of homeopathy clinical trials.34-35
Since the 1990’s, multiple meta-analyses of randomized placebo-controlled homeopathy clinical trials for any indication have been conducted with published findings.36-41 Hamre et al published a systematic review of six meta-analyses, two covering individualized homeopathy, one covering non-individualized homeopathy, and three covering both.42 These meta-analyses are composed of about 16 to 110 trials that were conducted between 1943 and 2014. Five of six meta-analyses found a significant positive effect of homeopathy over placebo. After conducting sensitivity analyses using only high-quality trials, four analyses remained, three of which continued to show statistically significant benefits (two in both individualized and non-individualized homeopathy, and one in individualized homeopathy), while one meta-analysis of non-individualized homeopathy no longer demonstrated significance.42
The observed small but statistically significant overall effect of homeopathy in well-designed meta-analyses serve as a signal warranting further investigation through intentional study design focusing on individualized homeopathic treatments. The American Institute of Homeopathy promotes awareness of research by offering guides, webinars from leading scientists, and an updated compilation of evidence-based homeopathy studies.43-44
Safety and Drug Interactions
From a safety standpoint, adverse effects of homeopathic medicines have been evaluated in a systematic review to include 41 observational studies comparing homeopathy to control. Adverse effects from the homeopathy groups were classified as minor to moderate similar to the control groups according to the Common Terminology Criteria for Adverse Events (CTCAE). The homeopathy groups were associated with less adverse effects than conventional medicines or other complementary therapies.45
While oral homeopathic products are highly diluted and adhere to GMP standards of the HPUS, many products, especially topical preparations, have low dilutions containing high amounts of active ingredients. Although uncommon, some isolated case reports have documented adverse effects associated with homeopathic treatments.9 MedWatch is the FDA product safety program available for health practitioners, patients, and consumers to report adverse events for regulated drug products including homeopathic medicines.46
Patient Care Considerations
Since homeopathic products are largely marketed as OTC drugs, pharmacy professionals may be the first health care practitioners who patients or consumers turn to for questions and product information. In a survey conducted by Millward et al., practicing pharmacists indicated that their knowledge of homeopathy was limited, despite the widespread availability of HDPs for direct consumer purchase.47 Khader et al. found that faculty from U.S. pharmacy schools believe that pharmacists need to be knowledgeable about homeopathic products and be able to communicate this information effectively to patients.48
Pharmacists are trained to evaluate self-limiting conditions and to determine whether self-care is suitable or if referral for additional medical evaluation is necessary. When patients opt for alternative or complementary therapies, pharmacists should be prepared to identify situations when homeopathy is suitable for self-care and when it is not. Patients should be informed that homeopathic medicines may be used as complementary therapies for certain self-limiting conditions, if desired, but they are not FDA-approved to treat or cure disease. For example, patients should avoid using certain products called “homeopathic immunizations” as substitutes for conventional vaccines because the U.S. Centers for Disease Control and Prevention indicates that their effectiveness is not supported by reliable scientific evidence.13
As with any medication, patients should be educated on when to seek medical care and to avoid delaying or replacing needed medical treatments. Patients should stop using homeopathic products or consult a health care professional if their symptoms worsen, allergic reactions occur (e.g. rash, hives, swelling of lips, tongue, throat or eyes, nausea, vomiting, diarrhea, or wheezing), or if their condition does not improve. Patients with a known allergy to the homeopathic sourced ingredient should avoid its use. Although many homeopathic products sold in the U.S. are highly diluted, patients should still notify their health care providers about all drugs which they are using, including homeopathic medicines, to ensure that any potential drug interactions are properly assessed.13
Patients and consumers should be advised to carefully review product labels to identify homeopathic products, usually marked with the designation “Homeopathic”.27 Oral pellets are administered sublingually by allowing them to dissolve under the tongue. Oral liquids should be taken by mouth using the measuring device provided with the homeopathic product to ensure accurate dosing. Topical preparations are intended for external use only and should be applied to the affected area, avoiding open wounds and the eyes or mouth.
Patients should be advised to choose HDPs monographed with the official “HPUS” quality designation, encourage them to thoroughly read product labels, and advise against discontinuing or delaying prescribed therapies without first consulting their health care provider.
Educational Resources
The internet has provided virtually unlimited access to information for anyone who wants to learn about homeopathy. A recent national survey found that more than half of American adults seek health information online, including through social media platforms and mobile health apps.49 Pharmacy professionals should utilize reputable online resources to help patients and consumers with point-of-care homeopathic product selection, if desired, and patient consultations regarding their proper use.
Homeopathic organizations are available to provide education and online resources to the public and health practitioners. The National Center for Homeopathy offers online resources and virtual webinars to educate the public and raise awareness about improving health through homeopathy. Its website features a “Find a Remedy” tool that guides users in identifying homeopathic options for various self-limiting conditions, including those that affect pets.50 Additionally, manufacturers’ websites and mobile apps are available to help consumers find homeopathic medicines for their self-limiting conditions, if desired.22-23
Other organizations such as the Center for Education and Development of Clinical Homeopathy, the American Institute of Homeopathy, and the Homeopathy Research Institute have educational and research resources for health care practitioners.51-53 The Materia Medica is a reference book which provides information on specific homeopathic medicines and is indexed in alphabetical order by name.15 This article provides an overview of homeopathy. Pharmacy professionals are encouraged to review focused materials by therapeutic area such as Homeopathy for Oncologic Palliative Care.54
Conclusion
Homeopathic medicines are complementary therapies available on the market as primarily OTC products. An individual’s choice to use homeopathic treatment is a personal approach to health and wellness. When patients or consumers ask about products like Arnica montana, Alium cepa, Ignatia amara, or Nux vomica, pharmacy professionals should be prepared to provide accurate information and counsel them appropriately to promote the safe use of these complementary therapies.